What does it take to make it?
 ARTBIO / Radforsk
ARTBIO / Radforsk
What does it take to make it for new biotech companies? We are looking for answers, digging into the legacy and environment surrounding successful start-up companies like ARTBIO.
One of the newest members of Oslo Cancer Cluster is the clinical-stage radiopharmaceutical company ARTBIO. On their webpage, they present the company in these words:
ARTBIO is redefining cancer care by developing a new class of alpha radioligand therapies (ART) and building the ecosystem that maximizes their potential.
We first wrote about ARTBIO in an article presenting the newest members in January. Here, we can read that ARTBIO is shaped by a long-standing scientific legacy with nearly a century of pioneering work in radiation therapy conducted at the University of Oslo and Norway’s Radium Hospital.
What is this legacy all about, and what kind of environment is optimal for successful start-up companies like ARTBIO to grow up in?
The answer appears to be fourfold.
The obvious part: financing
Part of the answer for ARTBIO is the tremendous financing round that raised USD 90 Million in December last year, for progress in the pipeline and isotope technology development for a new class of alpha radioligand therapies. The news of the company raising this amount from private investors made headlines in the Norwegian newspaper E24. How was this possible in today’s slow and skeptical market?
“There are several reasons why we managed to raise one billion NOK in the series A investment. The first one is that ARTBIO is a very attractive investment case with a differentiated technology and pipeline within radio pharmacy, which is a field that is receiving a lot of attention internationally”, said Anders Tuv.
He is Managing Director at Radforsk, an early-stage evergreen fund dedicated to oncology. Radforsk is one of ARTBIO’s founders.
In the latest investment round Boston-based venture capital fund Third Rock Ventures was the main investor, together with an undisclosed healthcare fund and existing seed lead investors F-Prime Capital and Omega Funds, according to a press release from the company.
The crucial part: the right people
“Even though it is crucial to have the right technology as a foundation, it is a must to have the right people and teams. And on that note, we have hit the bull’s eye with ARTBIO”, Tuv said and added:
“The company has been rigged the right way to see it through and succeed with realizing their potential in a global competition.”
Tuv especially mentioned the hiring of ARTBIO CEO Emanuele Ostuni as a key success factor, who was also the first full-time employee. He is described as bright, tenacious, and with a compassionate drive to succeed for patients.
To the question what do you think it will take to get to the next milestone? Ostuni answered:
“Our goals all involve doing things that have not been done before. As such they require creativity, collaboration, and practical optimism.”
Ostuni added:
“We are working to start a phase 1 trial this year. That will require a working technology for P212 isolation and a robust process for manufacturing the therapeutic product. We are also working to create a broader pipeline of programs that addresses patient needs – several of these programs should also move forward this year.”
The historical part: the legacy
So far, we have touched upon the company’s technology, the people, and the financing. What about this long-standing scientific legacy that we started with?
Anders Tuv explained:
“The scientific founders behind ARTBIO are the same people that developed the only alpha-emitting radionuclide treatment on the market so far (Xofigo), the legendary researchers and founders Roy Larsen and Øyvind Bruland.”
Tuv added:
“I think that both the seed round and the series A round in particular have shown that we have worked hard and done many things right with ARTBIO. We got international specialist investors and founding CEO Emanuele involved at a very early stage with Roy Larsen and Øyvind Bruland, and F-Prime and Radforsk have been active investors, building the company and heavily involved from the beginning. We have been thinking globally from the start and extracted talent where they were to be found.”
This is the reason why ARTBIO has offices in Boston, Basel, London, and Oslo.
The part of the environment
Oslo Cancer Cluster Innovation Park and Incubator, and the entire Campus around the Radium Hospital, including Radforsk, offer an environment for companies like ARTBIO to begin their journey, especially in radiopharmacy and precision medicine in cancer.
“We have a strong legacy and a good ecosystem for radiopharmacy in Oslo, including the Radium Hospital and the environment in Oslo Cancer Cluster”, Tuv said, and Ostuni agreed:
“We appreciate the availability of flexible space that is already partially equipped at Oslo Cancer Cluster Incubator – it allowed us to get started quickly when we were building the company.”
“The culture of openness and support at the Radium hospital also made sure that we could establish collaborations with academic groups and progress some programs while at the same time providing educational opportunities for female scientists”, said Ostuni.
So there you have it; the somewhat complex answer to what it takes to make it – at least through the first serious financing rounds.
About ARTBIO and Radforsk
ARTBIO’s approach: Alpha radioligand therapeutics (ARTs) are gaining attention as a promising experimental modality for delivering lethal radioactivity directly to cancer cells. The unique ARTBIO approach selects the optimal alpha-precursor isotope (Pb-212) and tumour-specific targets to create therapeutics with the potential for the highest efficacy and safety. ARTBIO is currently advancing multiple pipeline programs with lead program AB001 first in human trials.
Radforsk is engaged in the commercialisation of cancer research. Our goal is to develop better cancer treatment – for all patients. We invest in and develop infrastructure through Radforsk Innovation and companies through Radforsk Invest. Radforsk Invest is an oncology focused investment fund dedicated to developing immunotherapies, precision medicine and radiopharmaceuticals. The fund has an evergreen structure allowing flexibility and focus on investments that will create long-term value. Radforsk Invest`s model is based on very active ownership as investors and hands-on company builders. Radforsk’s portfolio of companies spans from early start-ups to public companies with products on the global market. For more information, please contact Anders Tuv, Managing Director Radforsk Invest (at@radforsk.no).
The post What does it take to make it? first appeared on Oslo Cancer Cluster.
Advancing Cancer Research in Norway: Eli Lilly’s SUNRAY-01 Study
Eli Lilly selected Norway as the site for its groundbreaking study project, SUNRAY-01, despite the recent year’s decline in applications for clinical cancer trials.
This study examines the efficacy of the drug candidate LY3537982 on advanced non-small cell lung cancer (NSCLC) with a specific genetic alteration. and represents a significant advancement in health research and treatment for patients with KRAS G12C mutations, potentially reshaping the treatment landscape for this specific patient group.
The KRAS G12C mutation is a specific alteration in the KRAS gene, often associated with certain cancers such as non-small cell lung cancer. This mutation plays a significant role in promoting the growth and spread of cancer cells. Researchers are exploring methods to block or inhibit the effects of this mutation.
Decline in trails
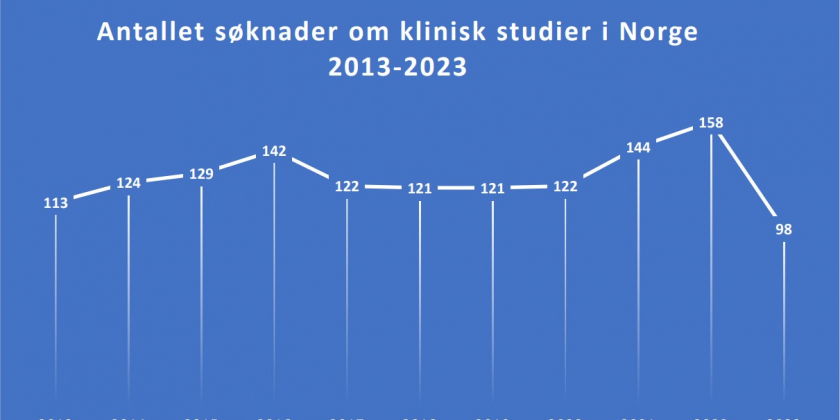
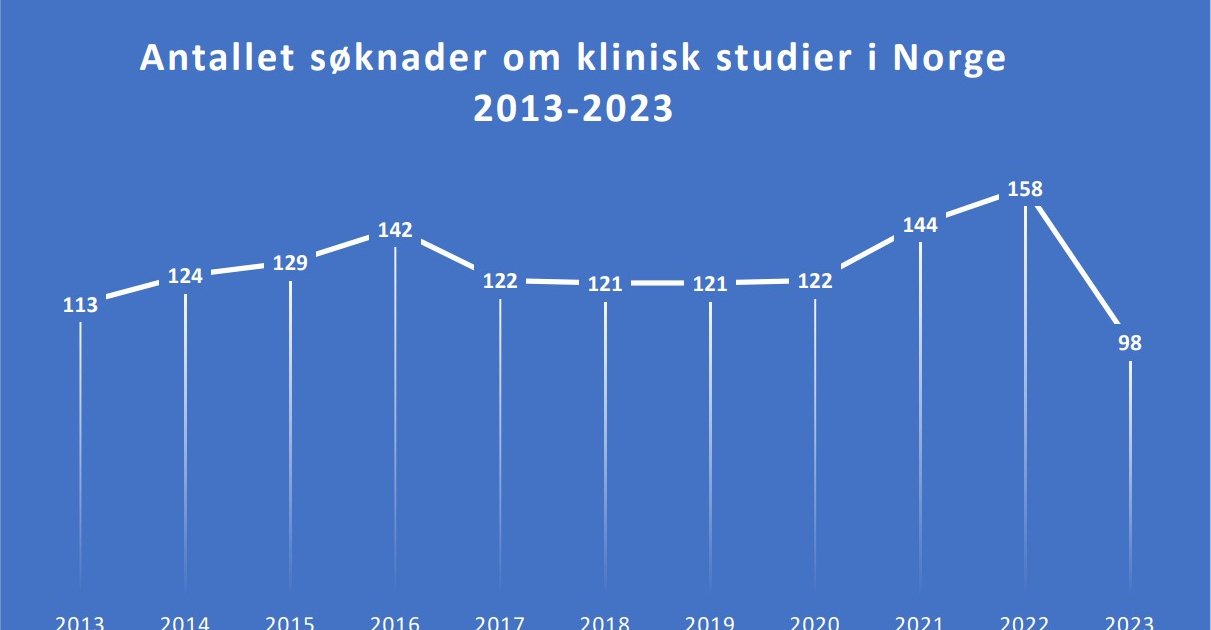
Recently, Norway has experienced a notable decline in the number of applications for cancer clinical trials, dropping from 158 in 2022 to 98 in 2023. Lars Petter Strand, Senior Medical Director for the Nordics at Eli Lilly, voiced concern, noting,

Lars Petter Strand. Photo Eli Lilly
“We observe that the number of cancer clinical trials in Norway has significantly decreased.” He highlighted the global trend of increasing clinical trials in countries like the USA and China, contrasting with the reductions in most European countries.
Norway’s participation in Eli Lilly’s SUNRAY-01 study indicates a positive shift. Lars Petter Strand attributed the decision to several favourable trends in Norway’s healthcare system, including initiatives like CONNECT, IMPRESS, InPRED, and NorTrials, which have enhanced infrastructure and processes, making Norway an appealing destination for clinical trials.
Positive outlook for patients
Bjørn Henning Grønberg, Head of Department for Translational Cancer Research at St. Olav Hospital, one of the 7 hospitals selected for this study, emphasized the importance of such studies, stating, “It is always welcome to offer study participation to our patients.” The proportion of lung cancer patients with KRAS mutations eligible for targeted treatment through this study exceeds those eligible for other targeted treatments.

One of the most exciting and significant aspects of this study is its focus on finding targeted treatments for KRAS mutations, which currently aren’t as effective as other options available.
Patients with this mutation respond to immunotherapy, unlike those with EGFR and ALK positives, making it an interesting combination to explore. However, in the past, this has been challenging, as the combination of KRAS inhibitors with immunotherapy was too toxic, says Grønberg.
Challenges and opportunities
Despite these positive developments, Norway encounters challenges in maintaining its attractiveness for clinical trials. Strand emphasized the importance of addressing barriers such as delayed introduction of new treatments, lengthy approval processes, and capacity constraints in diagnostic tools at hospitals.
The roadmap for the health sector, a strategic document guiding sector development, underscores the significance of clinical trials in health research. While the government has set ambitious goals for increasing clinical trials, collaboration across sectors and collective efforts are essential to address challenges hindering this vital part of medical research.
A roadmap for the health industry
Oslo Cancer Cluster General Manager Ketil Widerberg emphasizes that this new study aligns well with the Norwegian government’s aspirations for a national health industry and ongoing efforts at Oslo Cancer Cluster to foster innovation and collaboration within the cancer research field. It represents a crucial step towards advancing cancer care and supporting Norway’s health industry growth.
Widerberg stresses the importance of patients accessing the latest treatment, doctors and researchers gaining insights into the latest technology, and the development of the Norwegian health industry, as Norwegian centres of expertise gain international visibility.
Crucial collaborations
To attract more clinical trials to Norway, stakeholders must collaborate effectively, as Lars Petter Strand highlights. It requires creating sufficient resources in hospitals, facilitating efficient communication between the pharmaceutical industry and healthcare institutions, and streamlining startup processes. Improved communication between the pharmaceutical industry and hospitals is essential, as demonstrated by Eli Lilly’s collaboration with NorTrials during site recruitment for this study.
Collaboration between industry players, research institutions, and government bodies is crucial for advancing cancer research. Initiatives like NorTrials facilitate this collaboration, ensuring nationwide access to cutting-edge treatments, says Strand
The post Advancing Cancer Research in Norway: Eli Lilly’s SUNRAY-01 Study first appeared on Oslo Cancer Cluster.
Unique collaboration to build Nordic health industry

A new collaboration agreement between AstraZeneca, Oslo Cancer Cluster, and Oslo Science City aims to strengthen the Nordic health industry by facilitating closer collaboration between researchers, startups, and pharmaceutical companies.
This is a translation of an article in Norwegian, written jointly by Oslo Science City and Oslo Cancer Cluster. The Norwegian version can be read on Oslo Science City’s webpage.
The focus on investment in the health industry in Norway gained momentum after Minister of Trade and Industry, Jan Christian Vestre announced at the Norway Life Science conference in February 2023 that the government would develop a roadmap for the health industry.
In June, the health industry was selected as the fourth national export promotion initiative under the export reform “All of Norway Exports.” Two months later, Vestre and Minister of Health and Care Services Ingvild Kjerkol presented the roadmap, outlining 41 measures and 12 focus areas aimed at the sector developing drugs, medical devices, and digital tools.
Ahead of this year’s Norway Life Science conference, the Swedish pharmaceutical company AstraZeneca, Oslo Cancer Cluster, and the innovation district Oslo Science City are following up with a collaboration agreement to strengthen contacts and cooperation between Norwegian and Swedish research and innovation environments. The agreement will facilitate Norwegian startups’ access to residency at AstraZeneca’s innovation hub, BioVentureHub, in Gothenburg. Simultaneously, Swedish companies will have the opportunity to reside at Oslo Cancer Cluster and collaborate closely with their environments in cancer and precision medicine, as well as the outstanding research groups from the Radium Hospital, part of Oslo University Hospital, and the University of Oslo, which are gathered in Oslo Science City.
“It is important for AstraZeneca to contribute to the success of new startups and to strengthen the entire Nordic health industry. Therefore, we are very happy to participate in this collaboration, which will benefit all parties and build the Nordic region`s position internationally as a leading region in health and life sciences.” Guro Bjøntegaard, Managing Director of AstraZeneca Norway.
Industrial expertise and international connections
BioVentureHub was established in 2014 as an open and internationally oriented innovation ecosystem based on a public-private partnership model. Here, promising startups and research groups in Life Sciences can access office and laboratory space close to AstraZeneca’s strong professional communities and advanced research infrastructure.

AstraZeneca BioVentureHub is a not-for-profit innovation hub offering an inside track to AstraZeneca’s scientific expertise and facilities, for academic groups and small and medium-sized enterprises. It is integrated at the heart of AstraZeneca’s R&D center, in the new emerging life science ecosystem, in Gothenburg. Photo: AstraZeneca
“For our company, it is crucial to maintain close contact with the companies and knowledge communities that contribute to pushing the knowledge frontier in the fields we work in. Some of these companies and environments end up entering formal collaborations with AstraZeneca, but the most important purpose of BioVentureHub is to create a professional meeting place for creativity and innovation,” said Bjøntegaard.
From the Norwegian side, Oslo Cancer Cluster will have the role of identifying the companies that are offered residency in BioVentureHub. General manager Ketil Widerberg points out that promising Norwegian startups often lack industrial expertise and an important link to the international market:
“International collaboration is essential to scale up Norwegian startups. BioVentureHub can connect our start-ups to international value chains, and it is only a short train ride from Oslo. I think that will be quite effective!” Ketil Widerberg, Oslo Cancer Cluster
Lifting the entire Nordic region
The parties to the agreement also want to involve Innovation Norway, whereby Norwegian companies staying at BioVentureHub can apply for support during their residency. In the long term, the goal is also to involve Vinnova, Innovation Norway’s Swedish sister organization, in the collaboration.
“Health technology is an important Norwegian export industry, and at Innovation Norway, we encourage this type of broad Nordic collaboration between industry, startups, and research environments. By building stronger collaboration between the governmental agencies responsible for grants and support in the Nordic countries, we can contribute to lifting the entire Nordic region.” Håkon Haugli, CEO of Innovation Norway.
Over several decades, Norway has invested significant public funds in health research, but Christine Wergeland Sørbye, Managing Director of Oslo Science City, points out that several analyses show that we have been less successful than other countries in using this research to develop new companies.
“There is great potential here for business development that will both create new jobs and benefit Norwegian patients in the form of new medicines and treatments.” Christine Wergeland Sørbye, Oslo Science City
“By strengthening the collaboration between research and business across Nordic borders, we shall realize this potential,” says Wergeland Sørbye.
- Read more about the roadmap for the Norwegian health industry on the government webpage regjeringen.no (in Norwegian).
- More about the conference Norway Life Science is on the conference webpage.
The post Unique collaboration to build Nordic health industry first appeared on Oslo Cancer Cluster.